What is articular cartilage ?
To visualize a cartilaginous tissue, see the pictures below. The general structure of articular cartilage
is described hereafter under normal physiological conditions. Understanding this structure is a prerequiste to eventually
understand osteoarthritis. The approaches to understand osteoarthritis are necessarily interdisciplinary and
require multi-scales descriptions from the perspective of Physics, Chemistry, Physiology and Immunology. We start from the macroscopic view
to microscopic and nanoscopic views. Complex interactions with cell lines also occur at the different scales and will be touched upon.
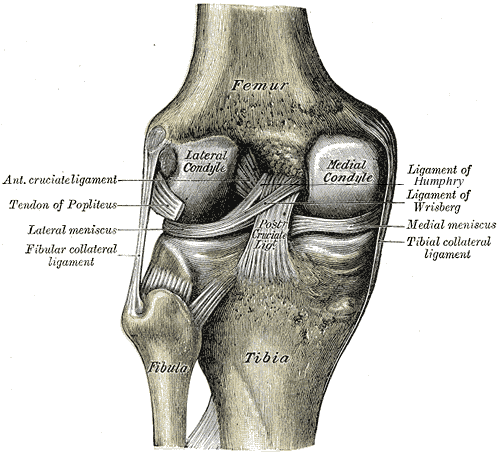
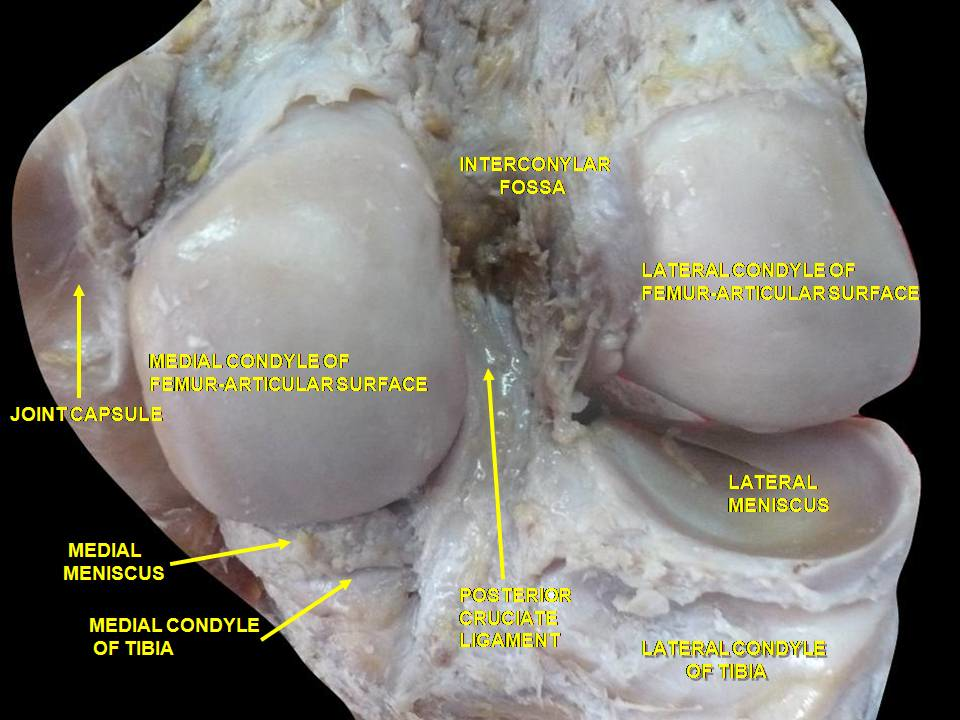
The bony surfaces of all freely moving adult synovial joints are protected by a thin, translucent layer of
connective tissue, known as hyaline articular cartilage. Approximately 0.5-7 mm thick, the key
components of articular cartilage are:
- Water which comprises 65-80 percent of its weight;
- Reinforced collagen (type 2 mainly) which comprises 10-30 percent of its weight;
- Proteoglycans which comprises 5-10 percent of its weight;
- Cells known as chondrocytes which comprises 1-10% percent of the cartilage volume;
- Small amounts of non collagen proteins (most of them belonging to the family of so
called Small Leucine Rich Proteins, i.e. SLRPs like decorin, biglycan, chondroadherin, ...), glycoproteins and lipids;
In terms of structural organization, chondrocytes, maintain the matrix components under normal, low turnover conditions.
The extracellular matrix half-lifetime is estimated to be in the order of 5-10 decades.
Osteoarthritis defined
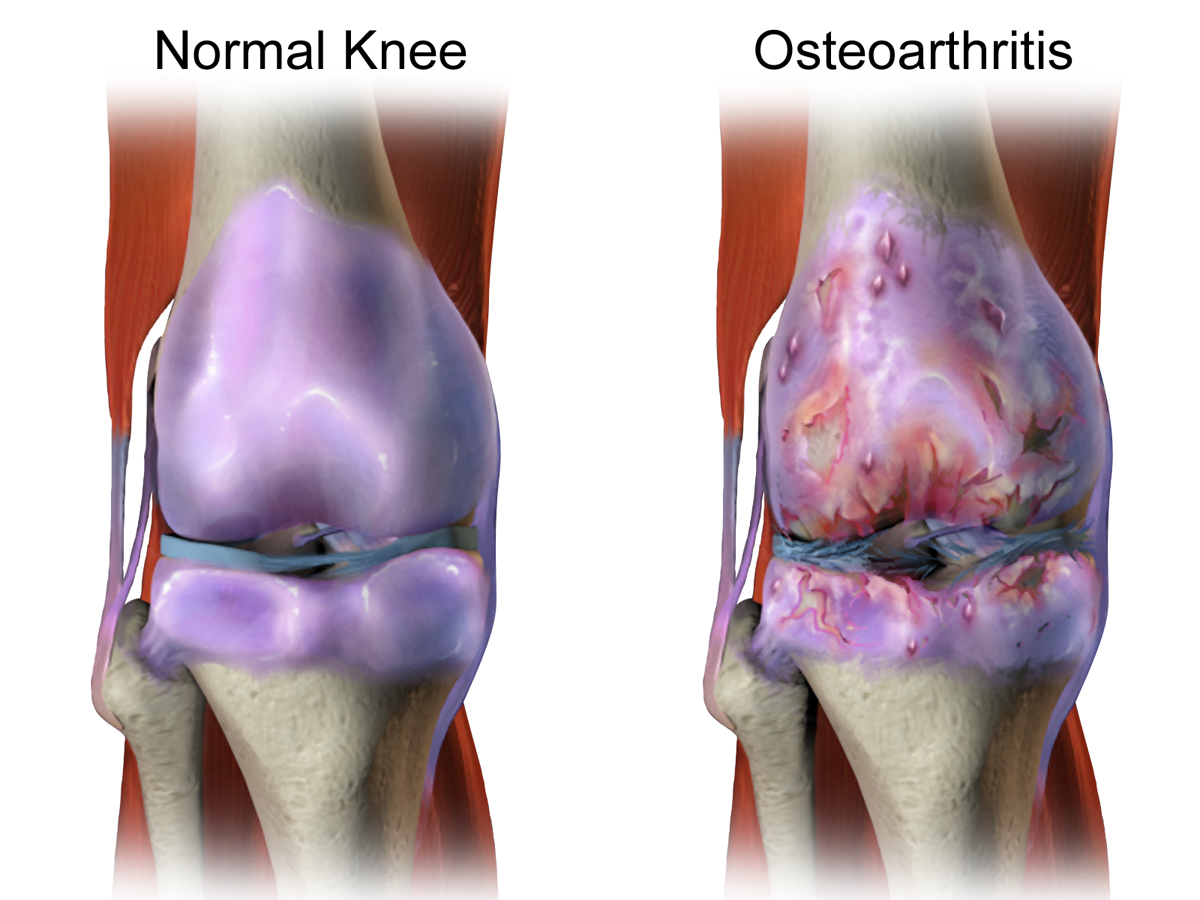
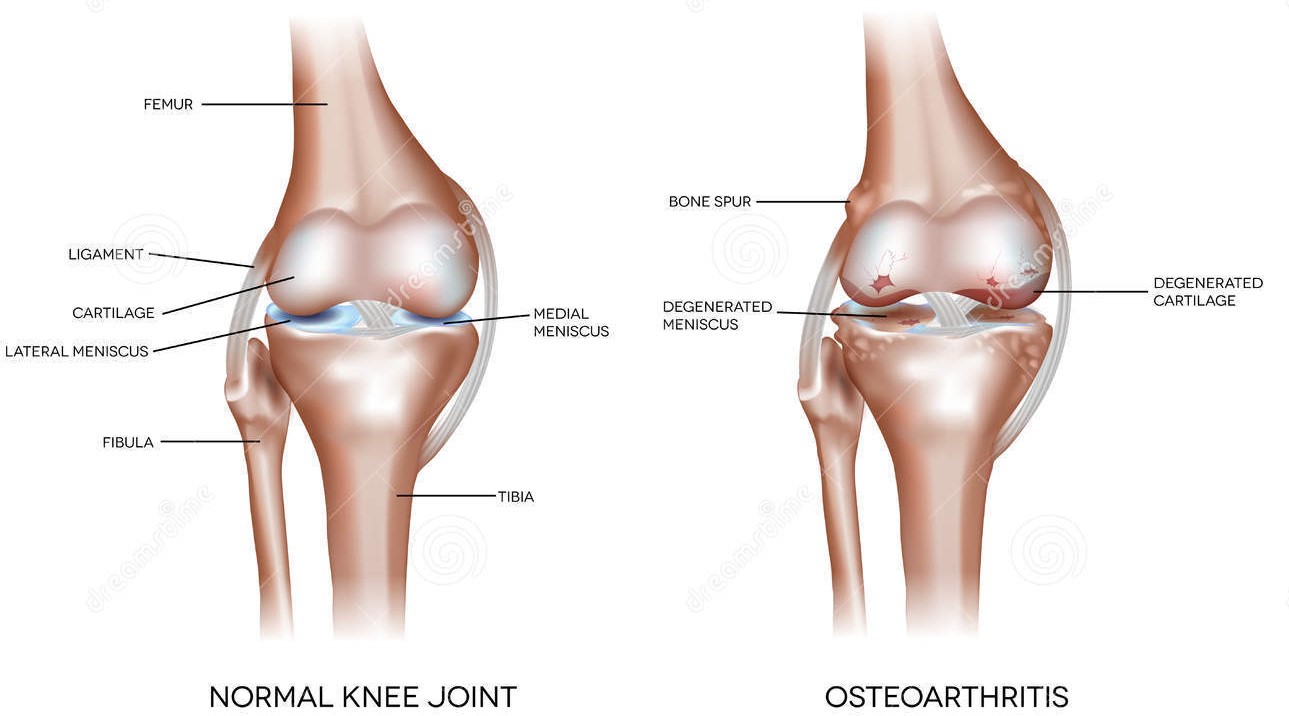
Osteoarthritis (OA) is a complex, multifactorial, age-dependent degenerative disease of the synovial joints. It affects
females at a higher rate than males, particularly after the menopause. OA is characterized by changes to all the
components of the joint, with degeneration and loss of articular cartilage and changes to the subchondral
bone being constant factors in disease progression. Along with the breakdown of the cartilage and joint space
narrowing, there is thickening and sclerosis of the subchondral bone, development of cysts and bony outgrowth at
the margins of the joint. Despite an increase in bone volume fraction, the subchondral bone is mechanically weaker
in OA because of hypomineralization, increased collagen metabolism and altered bone remodeling.
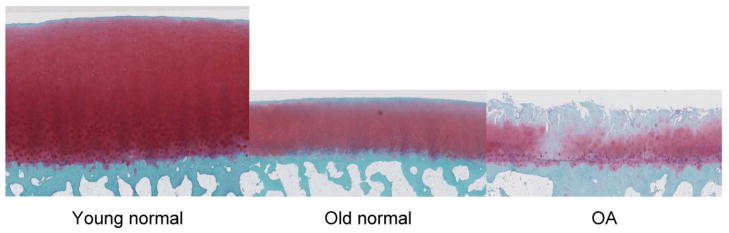
The pictures below illustrate differences in histological characteristics between human healthy young cartilage, older cartilage and
osteoarthritis affected cartilage.
There is a grading system for the assessment of osteoarthritis cartilage histopathology that is important in clinical practice and
called the Osteoarthritis Research Society International system (OARSI system).
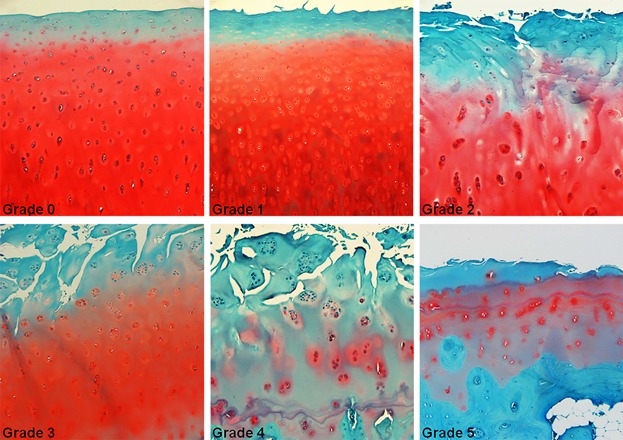
The figure illustrates the histologic characteristics of OARSI Grades 0 to 5. Grade 0: surface intact, cartilage morphology intact;
Grade 1: surface intact, superficial fibrillation, and edema or cell death or cell proliferation;
Grade 2: surface discontinuity; Grade 3: vertical fissures (clefts); Grade 4: erosion;
Grade 5: denudation. All pictures taken with a magnification × 100.
Credits: Waldstein, W. , Perino, G. , Gilbert, S. L., Maher, S. A., Windhager, R. and Boettner, F. (2016), OARSI
osteoarthritis cartilage histopathology assessment system: A biomechanical evaluation in the human knee.
J. Orthop. Res., 34: 135-140.
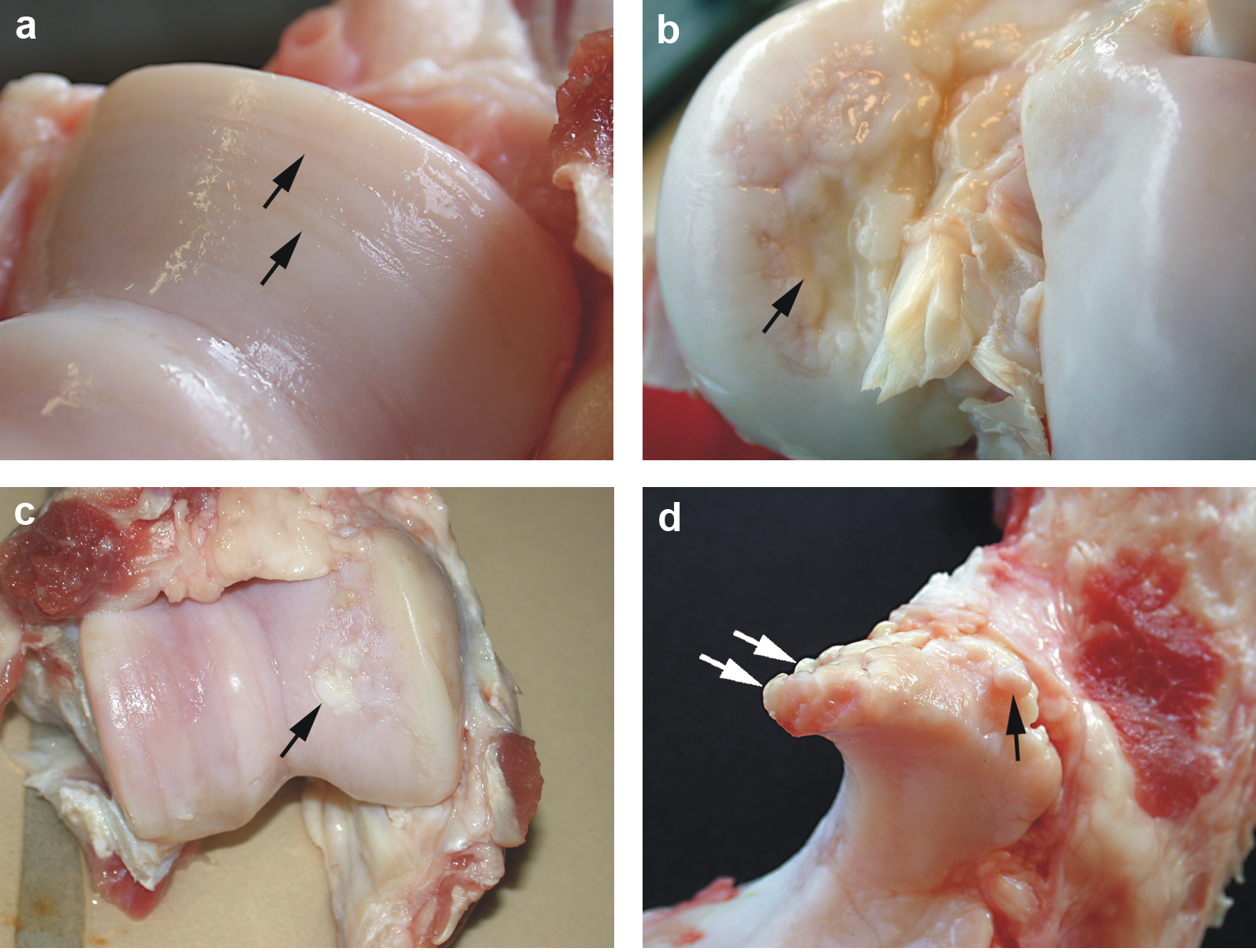
Damaged cartilages of a sow
The four pictures below show a dissection in a sow (female pig) cartilage. (a): Cartilage erosion (arrows) on the medial humeral condyle;
(b): Cartilage ulceration (arrow) on the medial femoral condyle; (c): Cartilage repair (arrow) of the medial femoral condyle;
(d): Marginal osteophytes (arrows) on processus anconeus of ulna.
Credits: Kirk et al. The impact of elbow and knee joint lesions on abnormal gait and posture of sows.
Acta Veterinaria Scandinavica. 2008;50:5.
The chemistry of the extra cellular matrix ECM
The extracellular component of cartilage consists of collagen fibers embedded in a gel
of glycosaminoglycans, proteoglycans and water, commonly referred to as the matrix.
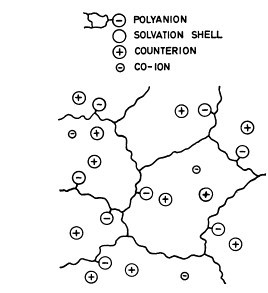
Following the pioneering work of Alice Maroudas back in 1968, this gel can be considered
as a typical ion exchange material, since it consists essentially of water and a network of linear polyanions
with fixed negative charges. The polyanionic matrix is indeed very similar to a strong acidic cationic (SAC) exchange resin.
The water is present partly in the form of solvation shells, and partly as free water with mobile cations
and anions in solution.
Sodium cations, the dominant counter ions present in the matrix, are attracted to the sulfates bound in chondroitin-sulfates
and keratan-sulfates glycosaminoglycans.
The high osmotic pressure that results has been measured and can be as high as 2.0 MPa, i.e. 20 atmospheres.
This chemical structure accounting for the high osmotic pressure explains how the extracellular matrix in
cartilage sustain mechanical stresses. Hydrostatic pressures higher than 2.0 Mpa should be applied before
the cartilage will start losing its water content.
Credits: Alice Maroudas. Physicochemical properties of cartilage in the light of ion exchange theory.
Biophys. J. 1968 May;8(5):575-595.
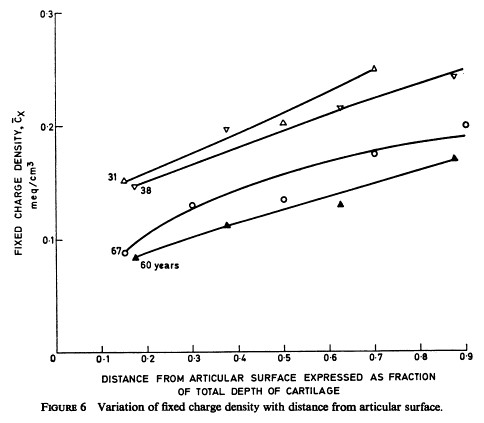
It is worth noticing that there is a fixed charge density gradient : the fixed charge density increases from the outer layers of cartilage to the inner layers closer to the subchondral bone.
As compared to men made artificial ion exchange resins like polystyrenic resins with sulfonate groups, the ECM matrix in
cartilage have a fixed charge density in the range 0.1 - 0.25 meq/mL, 5 to 10 times less charged than men made artificial resins (+/- 1.0 meq/mL).
Theoretically, the affinity of this ECM matrix in cartilage for calcium is very high and normally higher for
calcium than for sodium cations. If calcium ions were available in the ECM, the ECM would fix them preferentially to sodium ions.
Interestingly, the fixed charge density decreases with age as can been seen on the Fig 6 of the
original Maroudas pioneering paper reproduced here. The reason of this net charge density decline with age was not documented
in Maroudas' paper.
Collagens and glycosaminoglycans
The chemistry of glycosaminoglycans and open raised questions.
The basic units of NAG, N-acetylglucosamine and N-acetylgalactosamine.
How a complex structure such as aggrecan is built in the ECM from intracellular assemblies ?
The role of dolicholphosphate chemistry in the Golgi apparatus ?
How sulfatation occurs and where ?
Why the fixed negative charge density (sulfates) decrease from the subchondral region to the distal regions of cartilage ?
Cell lineages and crosstalks involved in cartilaginous tissues
Articular cartilage is an avascular, aneural and alymphatic connective tissue designed to distribute
mechnanical load and to provide wear-resistant surface to articulating joints. Chondrocytes are the
resident cells of cartilage and are the only cells found interspersed within the ECM. The chondrocytes
can synthesize all the components of this ECM. Despite their relatively homogeneous phenotype, chondrocytes
vary in size, morphology, and metabolic activity depending on which zone of articular cartilage they are found.
These specialized cells surround themselves with a terrotorial, pericellular ECM, primarily responsible
for mechanotransduction and an inter-territorial ECM that makes up the bulk of the load-bearing tissue.
When isolated and maintained in monolayer culture, these cells rapidly lose their fully differentiated functions
and de-differentiate into a fibroblastic phenotype. However, when they are grown in three-dimensional culture,
de-differentiated chondrocytes re-express the differentiated phenotype.
The interaction of chondrocytes with the ECM is mediated by the presence of transmembrane-receptors and proteoglycans,
namely, integrins and NG2 on their primary cilia which are involved in the signaling processes required for ECM synthesis
and maintenance. Consequently, normal cartilage matrix turnover is governed by chondrocytes, the rate of which is determined
by various stimuli.
For example, growth factors such as insulin growth factor-II (IGF-II) can stimulate DNA and GAG synthesis
in adult cartilage in vitro via their mitogenic effects on chondrocytes; whereas
cytokines such as interleukin-1\(\beta\)
(IL-1\(\beta\)) can cause cartilage degeneration by decreasing GAG synthesis by chondrocytes and/or
increasing the rate of GAG depletion.
Various intracellular and extracellular proteinases are thought to facilate this depletion, notably the metalloproteinases
(MMPs), which are responsible for collagen degradation, and the ADAM-TSs (A disintegrin and metalloproteinase with
thrombospondin motifs) which degrade aggrecan.
Thus the anabolic activities of chondrocytes are in dynamic equilibrium with their catabolic actions, maintaining the
homeostasis of the ECM of the articular cartilage. This balance can be affected by many factors including exercice and age.
Slight to moderate exercice can have chondroprotective effects via increasing the production of the anti-apoptic heat shock
protein 70 (Hsp70), although this protective effect is reduced as exercice intensity increases. Similarly,
increasing age reduces Hsp90 levels and their functional response to stress which is thought to play a role in the cellular
dysfunction observed with ageing. Using an Hsp90 inhibitor to stimulate reduced Hsp90 levels typical of ageing, it has been
shown that chondrocytes are less responsive to insulin-like growth factor-1 (IGF-1) and its anabolic effects such as cell
proliferation and collagen type II alpha 1 (COL2A1) gene upregulation. Thus ageing appears to disrupt the normal cellular
signaling pathways in articular cartilage.
Osteoclasts are of hematopoetic lineage.
Osteoblasts, osteocytes, chondrocytes.
Signaling pathways
Hormones, transcription factors, cytokines, interleukines, BMPs, TNFb, Wnt, Ihh and all that.
How mechanical stress or hydrostatic pressure or tensorial constrains are mechanotransducted
at the intracellular level and induce differential expression of genes in chondrocytes
or osteoblasts or osteoclasts ?
Biophysics and tensorial stresses
Mechanical and tensorial properties of cartilage.
How do we measure it ? How do you use it as covariate (explanatory variable) in the experimental settings ?
What is the link with osmotic pressure and the aggrecan-like structure ?
In vitro human cell culture of chondroprogenitors
Protocols for human cell cultures on scaffolds, hydrogels and the experimental settings.